Fixation
Purpose
To halt post-mortem tissue alterations and preserve cell structures in forms that resemble the living state. Different fixation methods are used according to the needs.
List of fixatives:
- Formalin, used for routine histology, but unsuitable for testes and embryos.
- Bouin's fluid, used for testes and embryos.
- Glutaraldehyde, used for transmission electron microscopy.
- Paraformaldehyde, preferred for immunohistochemistry experiments.
Methods of fixation:
- Immersion, the most classical.
- Perfusion, the most efficient. It is the quickest way to fix tissues and organs and is preferred for immunohistochemistry of organs that undergo rapid destruction (i.e., brain)
- Freezing preserves antigens, fats, RNAs and enzyme activity. It is used for certain immunohistochemistry, histoenzymology and in situ hybridisation experiments. However, morphological preservation is poor.
Equipment
- Automated printing system for embedding cassettes
- Peristaltic pump (perfusion)
Recommendations
Successful fixation by immersion requires small samples. The volume of the fixative should be 10 to 20 times the volume of the tissue samples.
Tissue processing, embedding and sectioning
Purpose
To embed fixed tissue specimens in a solid medium, which will support the production of thin slices destined for microscopic examination. The 5 steps in this process are dehydration, clearing, paraffin infiltration, paraffin embedding and sectioning. Bones are decalcified prior dehydration.
Equipment
- Automated vacuum tissue processor
- Paraffin embedding stations
- Microtomes
- Cryostats
Recommendations
After fixation, samples can be stored in dehydration step with 70% ethanol. They can not be stored in absolute ethanol more than 2 hours because this hardens the tissues. For the same reason, samples should not be stored in the clearing agent.
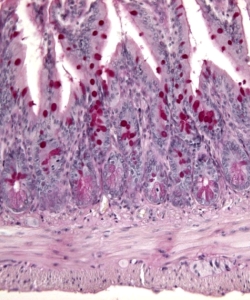
Staining
Purpose
To make cells and extracellular matrix visible. Routine histology uses the stain combination "hematoxylin and eosin". Other stains that bind selectively to specific tissue components include:
- Alizarin red and alcian blue for staining bones and cartilages
- Mallory's trichrome staining for fetuses
- Congo red stain for amyloid
- Von Kossa silver's nitrate method for detection of calcified tissues deposits
- Luxol fast blue and cresyl violet for staining of the central nervous system.
- Orcein stain for elastic fibers
- Oil Red O stain for neutral fats
- Periodic Acid Schiff (PAS) stain for glycogen
- Gomori's trichrome staining for hydrophobic struture of muscle (ie, mitochondria)
- NADH diaphorase staining for muscle
- Sirius Red stain for fibrillar collagen
- Masson's stain for fibrillar collagen
- Carmine-alum staining for mammary gland in toto
- Toluidine blue staining for mast cells
- Masson’s trichrome for fibroses
- Russell-Movat’s pentrachrome staining for collagen, elastic fibres, muscle, mucin and fibrin
- TRAP staining for osteoclasts
Equipment
- Automated printing system for microscopic slides
- Automated slide stainer
- Automated coverslipper
Recommendations
Specific conditions of fixation and embedding are required for each special stain.
Immunostaining
Purpose
To localize specific
proteins on tissue sections by the use of labeled antibody as specific reagents
through antigen-antibody complexes, these are visualized by a marker such as
fluorescent dye or enzymatic activity labeling (i.e. peroxidase or phosphatase
alkaline activity).Since immunohistochemistry
involves specific antigen-antibody reaction, it has become a crucial technique widely
used in research laboratories as well as in clinical diagnostics.
The service includes:
- Use
of standard techniques for antigen visualization using either fluorochrome or
enzymatic detection systems on either cryosections or paraffin sections
- Established
immunostainings are already available, but specific protocols provided by users
are welcome, as well.
- Setting
up of new protocols using commercial antibodies: various fixation methods, antigen retrieving
methods and detection systems are evaluated to validate a robust staining
protocol.
Example
of available immunohistochemical protocols :
- Immunodetection of KI67, BRDU and anti-phosphohistone H3 on organ sections to
evaluate proliferation (see proliferation and apoptosis)
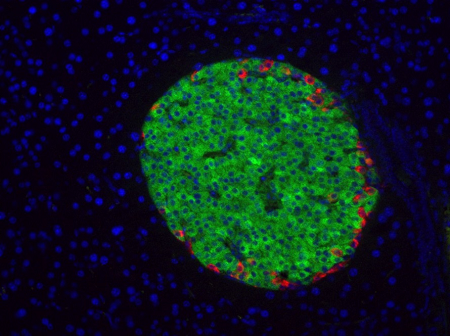
- Double immunostaining with insulin and glucagon on mouse pancreas sections to evaluate the islets of Langerhans
- Triple
immunodetection of glucagon, insulin and
somatostatine on pancreas sections to evaluate the islets of Langerhans
- Immunodetection of collagen 1 on heart sections to evaluate fibrosis
- Immunodetection of ANP and BNP on heart sections to evaluate cardiac diseases
- Immunodetection of adipophilin on organ sections to evaluate the presence of lipid
droplets
- Immunodetection of tryptase on organ sections
to evaluate mast cells.
- Immunodetection of Anti-neutrophil granulocytes (NIMP-R14) on mouse tissues
- Immunodetection of Anti-CD31 (blood vessels) on mouse tissues
Deliverables
Organs in paraffin blocks
- Fixation : 10% formaldehyde, Bouin's fluid, 4% paraformaldehyde or glutaraldehyde
- Decalcification : DC3 or EDTA
Histological sections from paraffin-embedded or frozen tissue blocs
- Unstained slides
- Hematoxylin & eosin- stained sections
- Special stains